INTRODUCTION
Trifluoroacetic acid (TFA), is widely used in organic synthesis as a solvent, catalyst and reagent thanks to its reactivity and stability under adverse conditions.
It is considered one of the most economical fluorinated building blocks. It is widely used in peptide synthesis and other organic transformations involving deprotection of t-BOC group.
Manufacture
Trifluoroacetic acid is commonly produced in Simon electro-fluorination process involving hydrogen fluoride and acetic acid.
| Synonyms | Trifluoroethanoic acid Perfluoroacetic acid |
| CAS no. | 76-05-1 |
| EINECS no. | 200-929-3 |
| Molecular formula | C2HF3O2 |
| Molecular weight | 114.02 |
| Structure | 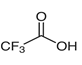 |
General Usage
The acid owes its multiple usages to its low boiling point, high dielectric constant, and relatively stronger acidity. It is generally employed in chemical industry as:
| Solvent in polymerization and oxidation |
| Solvent for chromatography |
| Catalyst in esterification, transesterification, and olefin reduction in petroleum refining |
| Low Boiling Reagent in various reactions |
Niche’ Applications
The chemical processes, that lead to value added products, are centered around the carboxylic group of trifluoroacetic acid and the stability rendered by the trifluoromethyl group. The special applications include:
| Trifluoromethylation – introduction of CF3 group in organic molecules thereby creating CF3 building blocks |
| Hydroacylation of complex organic molecules |
| Acylation of aromatics to form corresponding ketones and a plethora of acyl-containing intermediates |
| Olefin Hydration to produce alcohols – it replaces sulphuric acid, to which olefins are sensitive |
| Deprotection of amino group in some peptide synthesis for its higher solvency and volatility |
| Rearrangement – the acid is a key reagent in converting cyclohexanone oxime to ε-caprolactam |
| Protein Synthesis – where the acid protects the active amine |
Proxime Intermediates
The fluorinated acetic acid, by conventional synthetic processes, is easily converted to next step intermediates, that are vital for pharma and agro industries.
In these processes as well, the active carboxylic group and the stable trifluoromethyl moiety are the factors responsible.
These include:
| Trifluoroacetic anhydride – that further produces Thiazafluron |
| Trifluoroacetic acid ethyl ester (also known as ethyl trifluoroacetate) |
| Trifluoroacetic acid methyl ester |
| Trifluoroacetic acid isopropyl ester |
| 2,2,2-Trifluoroethanol – that is in turn consumed to make isoflurane and polythiazide |
High-End Intermediates
Trifluoroacetic acid finds applications as a key raw material to manufacture number of value-added, high-end intermediates such as:
| Peroxytrifluoroacetic acid |
| 2-(2,2,2-Trifluoroacetylamino)pyridine |
| N[1-{6-Chloro-3-pyridinyl)methyl)-2(1H)-pyridinylidene]-2,2,2, trifluoroacetamide |
| Methyl 2-Fluoroacrylate |
SPECIFICATIONS
| Test | Unit | Specification |
|---|---|---|
| Appearance | – | Clear & colorless liquid |
| Moisture | % w/w | 0.05 max |
| Fluoride | % w/w | 0.005 max |
| Chloride | % w/w | 0.001 max |
| Sulphate | % w/w | 0.001 max |
| Arsenic | ppm | NMT 8.0 |
| Titrimetric Purity | % w/w | 99.9 min |
| Identification by Ion Chromatography | The retention time of TFA matches to that of the standard TFA |
REACH status
TFA offered by ExSyn is registered under EU REACH regulations.
STORAGE
Product is stored at ambient temperature.
ExSyn offers this product on commercial scale and welcomes enquiries. No matter the quantity you need, our exceptional quality and service will make ExSyn your supplier of choice! If you need any additional information or SDS, please get in touch with us.
Sodium Hyaluronate is the sodium salt of hyaluronic acid, a naturally occurring glycosaminoglycan known for its exceptional ability to retain moisture, along with its viscoelastic and biocompatible properties, making it highly valuable across pharmaceutical, cosmetic, and biomedical applications.
Pyrazinamide is a first-line antitubercular drug used in combination therapy for the treatment of tuberculosis (TB). It is a synthetic pyrazine derivative and acts as a prodrug, which is converted into its active form (pyrazinoic acid) inside Mycobacterium tuberculosis.
6-Bromopyridine-2,3-diamine is a heterocyclic organic compound belonging to the class of substituted pyridines. It is used mainly as an intermediate in pharmaceuticals and agrochemicals.
Magnesium trisilicate is an inorganic compound commonly used in pharmaceutical and gastrointestinal (GI) preparations due to its dual action. It is usually produced as a white, odorless, fine powder. It helps bind toxins or irritants in the gastrointestinal tract and occasionally used in formulations for its stabilizing properties.
Choline salicylate is a salicylate salt formed from choline and salicylic acid. It is a non-steroidal anti-inflammatory agent (NSAID-type compound) mainly used for its analgesic, anti-inflammatory, and mild antiseptic properties. Unlike acetylated salicylates, it is less irritating to the gastric mucosa and is commonly used in topical oral preparations such as gels and mouth paints.
TensyraTM N-10 (Octoxinol 10; tert-octyl phenyl ethoxylate) is a non-ionic surfactant known for its excellent solubilizing and membrane-permeabilizing properties. It features a hydrophilic polyethylene oxide chain paired with a lipophilic aromatic hydrocarbon group.It readily breaks down biological membranes to lyse cells, solubilize proteins, and permeabilize cell membranes for assays.
Teprenone (Geranylgeranylacetone, GGA) is a mixture of the all-trans and mono-cis isomers of 6,10,14,18-tetramethyl-5,9,13,17-nonadecatetraen-2-one (in a weight ratio of 3:2). It is an acyclic isoprenoid compound with a structure like retinoids and is widely used for the treatment of gastrointestinal ulcers.
Ciclopirox Olamine (CPO) is the N-oxide of a 2 -hydroxy pyridine derivative that carries antifungal, antibacterial, and anti-inflammatory properties. It is composed of 6-cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridone combined with 2 aminoethanol (olamine) in a 1:1 ratio.
N,N-Dimethylglycine sodium salt (DMG Na) is the sodium salt form of N,N-dimethylglycine, a tertiary amino acid derivative is white to off white crystalline solid, freely soluble in water.